1 min read
Condition Monitoring – Don’t Have Stored Fuel Without It Part 5
This summer we hosted a webinar called Condition Monitoring for Stored Fuels. During the webinar we explained what condition monitoring is and the...
10 min read
Erik Bjornstad : Oct 12 2018

This summer we hosted a webinar called Condition Monitoring for Stored Fuels. During the webinar we explained what condition monitoring is and the key elements of condition monitoring, why it’s important, and how you should be doing it. You can watch the recording of the webinar here, or you can read the transcript in this five-part series.
Up to this point, we haven't really touched in detail on what happens as these microbes are growing and reproducing. What we mean by that is how do these microbes affect and interact with the environment around them and for a lot of you out there who have stored fuel, that means how are they affecting and how are they interacting with the storage tank environment and the fuel environment that they're in.
From a simple level, microbes as they grow and they reproduce, they produce things like called metabolites. That number of different kinds of metabolites have, should we say bad effects on fuel and storage tanks. There's a whole laundry list of kinds of things that they produce. They produce acidic components, alcohols, sulfate, salts. One thing of interest there is biosurfactants. A chemist might call them a zwitterion, which is a great Scrabble word. They produce these zwitterions, the biological surfactants. If you know anything about chemistry, then you know what a surfactant is. If you don't know anything about chemistry, the simple explanation for what a surfactant is it's a molecule that's got an electric charge on one end, which means it's attracted to what they call polar molecules, like water.

Water is a polar molecule, so one end of the surfactant molecule is going to be attracted to something like water. Then the other end is what they call non-polar because it doesn't have an electric charge and that makes it attracted to non-polar things, which luckily for us is precisely what the petroleum and fuel are. They are non-polar components. Now, if you've ever seen why are white bears attracted to each other, if you've ever seen, why do they attract each other in groups? Because they're polar, right? That was my one bad joke for the day.
What these biosurfactants do is they're produced by the microbes, and they actually help the microbes disperse water up into the fuel. Then the microbes will use that to help get at the food molecules in the fuel more easily. Of course, you can probably picture what that does to the fuels, what I just described. That's why when you have microbes; they're associated with hazy fuel because they produce biosurfactants that help emulsify water.
Speaking of emulsifying water and making fuel hazy and that kind of thing, now we’ve touched on something important. What other effects do microbes have on the fuel and the environment around them when they're producing these kinds of things? Well, generally speaking, the effect that microbes are going to have on different kinds of fuels depends on that kind of fuel that they're in.
Start with gasoline. Now for years, it was thought within the industry that microbes didn't really like to grow in gasoline. The reasoning that they used was that they thought that the lower end hydrocarbons, the C4s through the C7s, they thought that those components were actually toxic to a lot of different kinds of microbes. However, given enough time, they now realize that that's not really the case, that the traditional logic was faulty because contemporary research has shown pretty definitively that there are plenty of kinds of microbes out there that do like to live in gasoline and that they actually liked to consume those low-end hydrocarbons as fuel.
Of course, they also like to consume the oxygen that's now ever-present in gasoline. Now, how does that affect the gasoline? Well, if you think about it if the microbes over time consume a significant portion of the lower end molecules, what does that do? It changes the molecular distribution of the fuel, which means it changes the percentage of aromatics that are in the fuel blend. It can affect the specific gravity, it can affect the vapor pressure of the gasoline over time, and none of those are particularly good things that you want to have happening in stored gasoline. Of course, we know that store gasoline is not the biggest slice of the pie that we want to be concerned with. It's really stored diesel fuel that everyone's concerned about. What happens with that?
Well, because this fuel obviously is more likely to be stored for a longer period of time, it's a lot more common to find microbes and things like the associated water phases. It's a lot more common to find those in diesel storage tanks.
When you find them, what's likely to be happening there when you find them? Well, in terms of what microbes need to grow, of course we all know that microbes need some kind of water around to grow and multiply. Microbes can live, what they call planktonically in floating around in fuel, but in order to really grows and multiply substantially and form these kinds of colonies that end up causing the problems, they need some kind of free water, which isn't hard to find, but they do need it.

Now, that being said, they do not need very much of it in order to do what they need to do. Now, earlier we mentioned this thing about one-millimeter layer of biofilm is 100,000 times the size of a microbe. Now, consider if you find a water phase if you have just a one eight-inch water phase in the bottom of a tank. Now one eighth inches oftentimes too small to be detected by water phase but if you have one eighth inch water phase and you put a microbe, one little bacteria microbe in that and you look at the size of just that one eighth of an inch compared to that microbe, that is analogous to the size difference when a human being is standing at the foot of Mount Kilimanjaro and looking up at it.
Because microbes are so small, right? Reconcile that too with what we said earlier about one milliliter of water being enough to support between a million and a billion microbes in many cases. They need free water, but they don't need very much of it. When you get free water and microbes present under the fuel, the microbes are going to use that water in certain ways in order to do certain things. The first thing that's going to happen is that certain polar constituents that were in the fuel are going to start migrating into the water where the microbes will gladly receive them and use them in part for fuel. Then, as we alluded to earlier, the microbes start producing these biosurfactants things, and the biosurfactants take some of that water, and they emulsify it up into the fuel, and they create what they call an invert emulsion, which is a water-in-fuel oil emulsion.
Then the microbes then use that emulsion to migrate up into the fuel where they can now better get at the surrounding fuel molecules for fuel, and the process repeats and the microbes are very happy. Now, over time, what effect is that going to have on the storage tank?
Well, you can't really have a complete talk about microbes without examining the effects that the microbes have on the tanks and the environment that they're stored in. The two terms that are most often used when talking about the subject. We talk about fouling, and we talked about corrosion, right. Now, we're used to thinking of corrosion as a general descriptor of the bad things that microbes do to storage tanks, but that's not always entirely accurate because corrosion technically happens to metal. There's a lot of storage tanks that while they have metal components, they may be made out of fiberglass and they have obviously nonmetallic linings and a lot of nonmetallic components, and yet we still see a lot of damage that microbes can cause in those situations.
We would call that noncorrosive damage. What are some of the things that microbes do?
Well, of course, they produce this biomass slime and the slime, of course, accumulates non-uniformly within the tank, and sometimes it can foul the fuel filters, and it can also affect the tank floats. If it gets into the tank float, then it can actually change the floats specific gravity which causes it to give an inaccurate fuel volume reading. Now, what about with linings? Microbes definitely can damage tank linings. Over time, you'll get some kind of pin hole compromising, or just little pinhole in the lining and what happens then is that fungus-like, fungi like to do this, fungus along with additional moisture will actually migrate into that pinhole and will start to get underneath the lining.

Now over time, with temperature changes and the light, the moisture will expand and contract and that causes the lining to start to become debonded from the tank shell. Eventually, you'll get it; you'll see where you have blistering and peeling away from the shell. Now, sometimes in pretty large sheets if it's severe enough. Fungi can also compromise just regular fiberglass tank lining, tank surfaces as well. You know, a fungus doesn't necessarily need a pinhole to do this. It can colonize this fiberglass surface, and it will send up what they call the high phase, a little if you want to call them threads, but it'll send out little fillers that will penetrate into the surface of the fiberglass. Will penetrate the resin, and the fungi will start to consume the fiberglass resin. It will deplasticize the resin, create more gaps, which then more microbes and additional moisture can get in there, you get the whole expansion and contraction thing, you get little fractures that happen, and over time, you can get pretty severe compromising of even fiberglass by microbes.
So up to this point, we've talked about a lot of details about the microbes themselves. So I think we've talked enough about that. We now want to transition into talking about some of the corrosion mechanisms, and then we'll start to transition into actual techniques for, specifically, condition monitoring as well.
So, with corrosion; we just talked about non-corrosion damage. Now, with corrosion, there are a number of different mechanisms on how that can happen inside of a storage tank. The most prevalent mechanism for tank corrosion that's observed, what they call "in the wetted area," that means below the fuel line, as opposed to vapor space corrosion.
The most prevalent mechanism for tank corrosion in the wedded area involves the formation of galvanic couples that form between the steel of the tank and things like iron sulfides that are produced by the microbes. That's one of the reasons why sulfate-reducing bacteria are so bad for causing corrosion because they produce these iron sulfides and what happens is that you get movement of electrons between one kind of surface to another, which accelerates corrosion reactions.
Now, we talked earlier about biofilm in a number of different contexts. But a lot of people don't realize that in addition to being, you know, plugs filters and stuff like that, biofilms actually play a key role in helping corrosion damage to happen in tanks, which is something you wouldn't really think unless you start looking at it a little closer. It's one of the reasons why addressing, if you find biofilm and biomass in your tanks, you need to get it cleaned out. Because if you don't, it will cause corrosion in your tanks.
Well, how does that happen? Well, a number of different ways. The first way is that if you have some biomass that's present on a storage tank surface, then it will help create this electro potential gradient between the uncovered and the covered surface. You get a movement of electrons between one and other, between one surface to another, rather, creates pinhole leaks in the steel surface and further corrosion damage.
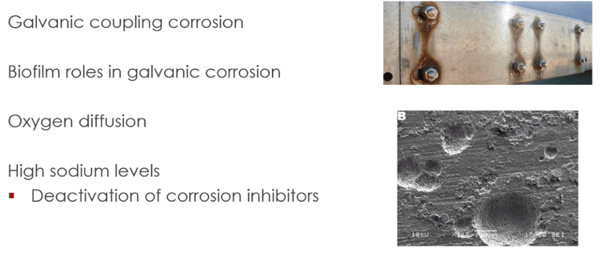
Another way that happens is that it interacts with oxygen. Now, this is interesting; oxygen, of course, plays a key role in the development of corrosion damage. Now, you may not think it, but fuel or petroleum actually dissolves more oxygen than water does. You can get up to, say, 60 parts per million of dissolved oxygen in a given kind of petroleum. Some people say up to ... 200 parts per million water, which has oxygen associated with it. But in terms of just O2, about 60 parts per million. Now, for water, that limits about eight. So, it's like, you know, 20% or 15%.
Now, what happens when you have such a difference in oxygen concentration between, when you have fuel, and you have a water phase that's next to it, what happens is that you have, of course, the principle of osmosis, right? So, as we learned in science class from years ago, osmosis is the tendency where things like to move from areas of high concentration to areas of low concentration, if they can. If there's a permeable or semi-permeable membrane or something that they can move across.
So what's going to happen if you have a layer of water and a layer of fuel, the fuel has more oxygen than the water, so what's going to happen is that some of that oxygen's going to want to migrate from the fuel into the water. Now, what happens if it's doing it right next to a biomass formation? Well, what will happen is that you will get some areas, some of that oxygen will get trapped in parts of that biofilm, and you will start to get little, localized areas where there's a high oxygen concentration. And that there produces localized corrosion and speeds up the corrosive metal loss. And it can be affected by things like temperature, the presence of hydrogen sulfide, the presence of certain kinds of acids and salts ...
When you talk about sodium, if you have sodium levels in the tank water that exceed like a tenth of a percent, that can actually affect or help contribute to corrosion in the tank, because what happens is that those actually interact with ... certain acidic components that the microbes have made, and they produce these things, these chemical soaps. Now what the soaps do is they interact with the chemical corrosion inhibitor additives that are already present in the fuel, and they actually inactivate those. So, when that happens, you've got a greater chance of corrosion to happen because the high sodium level has affected the ability of those corrosion inhibitors in the fuel to do what they need to do.
Now, what about the microbes themselves? What kinds of things do they do? Well, we've already talked about the fact that they produce certain kinds of things. And what, more specifically, they do depend on the kind of microbe that you're talking about. Now, if you have an acid-producing bacteria, an acid-producing bacteria will produce, what? They'll produce acids, of course. But they'll produce different kinds of acids. They can produce what they call these 'low molecular weight' acids, basically small molecule acids like ... methanoic acid, well, methanoic acid would be one. Acetic acid, of course, is a big one. But why that matters is that those are actually light enough to actually vaporize and go up into the vapor space in the tank.

Some of you might have been on a webinar we did last year where we talked about the EPA report on microbial-induced corrosion in storage tanks, and they talked a lot about this phenomenon of vapor space corrosion. And a lot of it was due to the fact that these lightweight acids actually travel up above the fuel line into the vapor space and cause significant corrosion damage up there.
And then there are of course other species that produce a lot stronger acids; they produce carbon dioxide which can combine with water to produce carbonic acid, which is highly corrosive to metal. So how quickly does this happen? And this is the last thing that we'll say before we transition into condition monitoring.
How quickly does this happen? Well, if you look in what they call the literature value, the average literature value for corrosion rates in storage tanks is between 0.3 to 0.5 millimeters per year. And that's for diesel tanks. Gasoline tanks tend to have slower corrosion rates, around 0.1, 0.13 millimeters per year. But there are instances where it has happened significantly faster than that.
Now, the last thing we'll say about this is part of the nature of the beast, so to speak, when we talk about the kinds of tank corrosion damage and fouling and microbial-induced corrosion that we've been talking about here, is that you rarely will find corrosion damage unless you're actually looking for it. Now, what does that mean? That means that once you actually look into your tank and you notice corrosion somewhere, once it's developed enough to the point that you notice it, the problem has been accelerated to where it's pretty serious. So basically, once you find it, you've got a serious problem. Kind of like if you're driving your car, once you're, you know, if your car pulls over to the side of the road, that's a serious problem. The problem had developed to the point that by the time you noticed it, it was too late, so to speak. And that's kind of how tank corrosion happens as well.
And so because of that, that is an excellent reason for us to look closer at the practice of condition monitoring in our fuel storage systems. And so that is what we're going to talk about next.
This is Condition Monitoring Part 2. You can read Part 1 here and Part 3 here.
Watch the full presentation below:

1 min read
This summer we hosted a webinar called Condition Monitoring for Stored Fuels. During the webinar we explained what condition monitoring is and the...

1 min read
This summer we hosted a webinar called Condition Monitoring for Stored Fuels. During the webinar we explained what condition monitoring is and the...

1 min read
This summer we hosted a webinar called Condition Monitoring for Stored Fuels. During the webinar we explained what condition monitoring is and the...